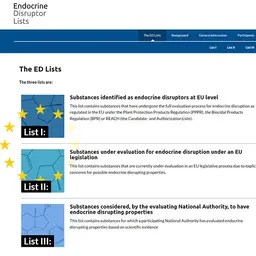
Over the past few months, the noose had been tightening around Methylisothiazolinone. It was banned in leave-on cosmetics as from 12 February 2017, and the maximum authorized concentration of this preservative should also be considerably reduced in rinse-off products by the end of the year. Here is a short reminder of regulations for formulators and companies placing products on the market.
0% in leave-on cosmetics
Regulation 2016/1198 of 22 July 2016 amended Annex V (preservatives) to Cosmetics Regulation 1223/2009, to ban Methylisothiazolinone in leave-on cosmetic products.
Grounds
In
an Opinion of 12 December 2013
, the SCCS concluded that current clinical data indicate that the concentration of 100ppm of methylisothiazolinone in cosmetic products is not safe for the consumer. For leave-on cosmetic products (including ‘wet wipes’), no safe concentrations of methylisothiazolinone for induction of contact allergy or elicitation have been adequately demonstrated.
Enforcement
Regulation 2016/1198 states that from 12 February 2017, Methylisothiazolinone is authorized only in leave-on products. Only compliant cosmetic products shall be placed and made available on the Union market.
0.0015% in rinse-off cosmetics
The draft regulation of the Commission notified to WTO on 28 November 2016 plans to amend Annex V to Regulation No 1223/2009 on cosmetic products and restrict the use of methylisothiazolinone as a preservative in rinse-off cosmetic products from the currently authorised concentration of 100ppm to 15ppm.
Grounds
In an
Opinion on the safety of Methylisothiazolinone
of 15 December 2015, the SCCS …