On 18 December 2025, the French ANSES published an article on its website explaining the reasons that led it to propose a CLH classification of sodium fluoride as ED HH 1 and Repr. 1B. The information was widely reported in the mainstream media, prompting FEBEA to issue a clarification.
Background
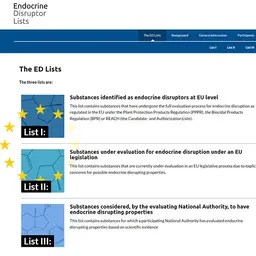
On 17 November 2025, the European Chemicals Agency (ECHA) launched a consultation on France’s proposal for the harmonised classification and labelling (CLH) of sodium fluoride, commonly used in oral care products, in particular as a reprotoxic substance and endocrine disruptor.
And in an article published on its website on 18 December, the Agency explains that: • Sodium fluoride had been identified as a substance to be assessed as a priority for its potential endocrine-disrupting properties under the second National Strategy on Endocrine Disruptors (SNPE2);
• Experimental studies in rodents and epidemiological studies in humans show that sodium fluoride has effects on neurodevelopment following exposure during pregnancy and that it alters thyroid function (effects observed following oral exposure). Furthermore, studies in animal models show impaired fertility.
In light of its assessment, ANSES has therefore proposed a harmonised classification of the substance, notably as:
• Repr. 1B, H360F - May damage fertility.
• ED HH 1, EUH380 - ED HH 1, EUH380 - May cause endocrine disruption in humans.
If this classification becomes effective, it could lead to a possible ban on the use of the substance in cosmetics…
The FEBEA’s response
The ANSES announcement was …