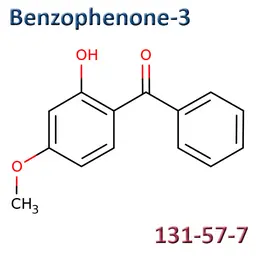
On October 15, 2025, the European Chemicals Agency (ECHA) announced Denmark’s intention to propose the harmonized classification and labeling (CLH) of the UV filter Oxybenzone (Benzophenone-3), as an endocrine disruptor.
Oxybenzone (CAS No.131-57-7, CE No.205-031-5, INCI: Benzopheneone-3) is an ingredient listed in the Cosing, with the functions: Light stabilizer, UB absorber, UV filter.
The substance is regulated by Annex VI/4 (UV Filters) of European Cosmetics Regulation 1223/2009 which limits its maximum concentration to 6% in face products, hand products, and lip products (excluding propellant and pump spray products), to 2.2% in body products (including propellant and pump spray products) and to 0.5% in other products.
It is not currently listed actuellement pas listée in Annex VI of European CLP Regulation 1272/2008.
Denmark intends to propose the following classifications.
• ED HH 1, EUH380 - May cause endocrine disruption in humans.
• ED ENV 1, EUH430 - May cause endocrine disruption in the environment.
Related Ingredient Datasheet
Sources
• ECHA Weekly - New intentions, proposals and a withdrawal to harmonise classification and labelling, ECHA, 15 October 2025
• Registry of CLH intentions until outcome - Oxybenzone, ECHA, 13 October 2025