Silicones are ubiquitous ingredients in the cosmetics industry, offering remarkable versatility thanks to their unique structure. However, they have been the subject of debate for many years, particularly with regard to their toxicity to humans and their environmental impact. So are they the sworn enemies of cosmetics, or simply the unloved ones who deserve a second chance? The point of view of Corinne Benoliel and Blandine Gutton, from Institut Scientis.
The main properties of silicones include exceptional stability, scalable hydrophobicity, chemical inertness and excellent skin tolerance. These characteristics make them prized ingredients in the world of cosmetics, from moisturizers and antiperspirants to make-up and hair care.
Chemical structure
Discovered in the early 20th century, silicones are synthetic compounds characterized by a unique structure based on siloxane (Si-O) bonds.
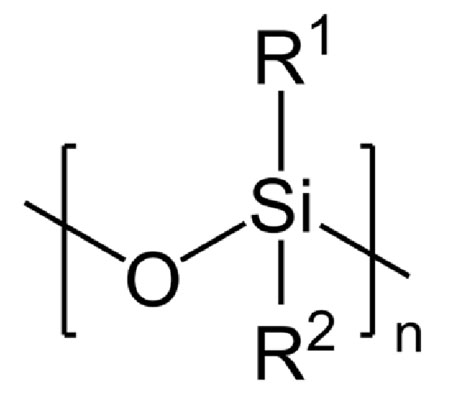
Various organic groups are attached to silicon atoms. This intrinsic organization gives silicones very specific properties, which explains their massive use in many sectors.
These substances can adopt different configurations: linear, cyclic, branched or cross-linked, responsible for diverse properties.
Dimethicone and Silesquioxane are among the silicones most commonly used in cosmetics and the chemical industry in general.
Properties
Silicones have many properties of interest to the cosmetics industry.
General properties
• Siloxane bonds provide stability and flexibility.
• The grafts present on siloxane structures modulate the compound’s affinity for water, generating modulable hydrophobicity or hydrophilicity.
For example, methyl groups make silicones hydrophobic, while polar groups such as polyethylene glycol (PEG) or polypropylene glycol (PPG) make them water-soluble.
• Silicones are chemically inert, i.e. resistant to oxidation, temperature, humidity, UV rays, …