Following their presentation at the 23rd Cosmetic Valley Regulatory Issues - Perfumes & Cosmetics Congress held on November 19 and 20, 2025, representatives from the DGCCRF answered questions from congress participants. Topics covered during this session included controls strategy, borderline products, claims, and GMPs.
Virginie Gallerand, Head of Office 5B, Nathalie Delion, Flore Pluvinage, and Isabelle van Hamme, Editors in charge of cosmetics at the DGCCRF, answered questions from conference attendees.
Controls
Question
How are you adapting your priorities and inspection methodologies to address new regulatory challenges such as those related to environmental claims, digital marketing, or the use of AI in product development and advertising?
Response from the DGCCRF
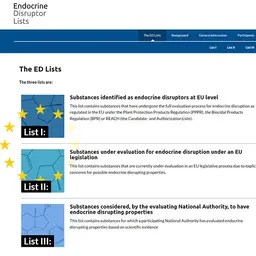
As mentioned in the presentation, the PNE (National Investigation Program) is based on a risk analysis that takes these new trends and emerging risks into account.
Question
Do you plan to begin expanded allergen inspections in July 2026 or after the end of the Regulation’s application period in 2028?
Response from the DGCCRF
As you know, Regulation 2023/1545 on the labeling of new allergens provides for entry into force on July 31, 2026, for products placed on the market and on July 31, 2028, for products made available on the market. These provisions will be taken into account during our inspections.
Question
Will you penalize service providers who do not perform any safety assessments on the primary packaging of finished products?
Response from …