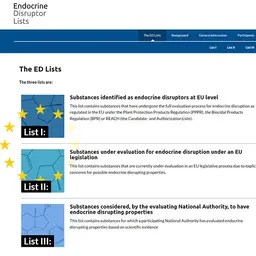
On December 3, 2025, ECHA (European Chemicals Agency) published Sweden’s proposal for the harmonized classification and labeling (CLH) of Silver Chloride, a preservative, in particular as a mutagenic, carcinogenic, and reprotoxic substance.
Silver chloride deposited on titanium dioxide (CAS No.7783-90-6, EC No.232-033-3, INCI: Silver Chloride) is an ingredient listed in Cosing, with the function: Preservative.
The substance is listed in Annex V/52 which limits its maximum concentration to 0.004% (as AgCl), and bans it in products for children under 3 years of age, in oral products and in eye and lip products.
It is not currently listed in Annex VI of the European CLP Regulation 1272/2008.
Sweden proposes the following classifications:
• Skin Sens. 1, H317 - May cause an allergic skin reaction.
• Muta. 2, H341 - Suspected of causing genetic defect.
• Carc. 2, H351 - Suspected of causing cancer.
• Repr. 1B, H360FD - May damage fertility. May damage the unborn child.
• STOT RE 1, H372 (nervous system) - Causes damage to organs through prolonged or repeated exposure (with specific concentration limits).
• Aquatic Acute 1, H400 - Very toxic to aquatic life. M-factor=1000.
• Aquatic Chronic 1, H410 - Very toxic to aquatic life with long lasting effects. M-factor=100.
If the process is completed, Silver Chloride could be listed as a CMR substance, and its use in cosmetic products in Europe could be prohibited, …