On March 4, 2026, the European Chemicals Agency (ECHA) announced Sweden’s intention to introduce harmonized classification and labeling (CLH) for Deltamethrin, an ingredient suspected of acting as an endocrine disruptor.
α-cyano-3-phenoxybenzyl [1R-[1α(S*),3α]]-3-(2,2-dibromovinyl)-2,2-dimethylcyclopropanecarboxylate (CAS No.52918-63-5, EC No.258-256-6, INCI: Deltamethrin) is an ingredient listed in Cosing, with no reported function.
The substance is currently not regulated by European Cosmetics Regulation 1223/2009.
It is already listed in Annex VI/607-319-00-X of European CLP Regulation 1272/2008, with the following classifications.
• Acute Tox. 3*, H301 - Toxic if swallowed.
• Acute Tox. 3*, H331 - Toxic if inhaled.
• Aquatic Acute 1, H400 - Very toxic to aquatic life (M-Factor = 1 000 000).
• Aquatic Chronic 1, H410 - Very toxic to aquatic life with long lasting effects.
In the CLH Registry of intentions, the substance entry indicates that the proposed classification is to be further specified.
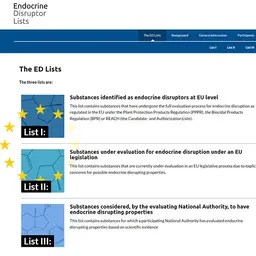
However, it is already known that it is suspected of acting as an endocrine disruptor, as it appears on the priority list of potential endocrine disruptors established by the European Commission in 2019.
Sources
• ECHA Weekly, Classification, Labelling and Packing (CLP) - New intentions and proposal to harmonise classification and labelling, ECHA 4 March 2026
• Registry of CLH intentions until outcome - α-cyano-3-phenoxybenzyl (1R-(1α(S*),3α))-3-(2,2-dibromovinyl)-2,2-dimethylcyclopropanecarboxylate, ECHA, 3 March 2026