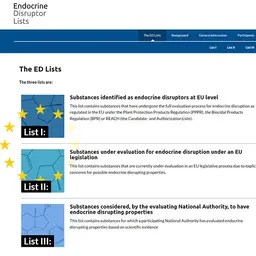
Following the classification of Hexyl Salicylate as a Category 2 CMR substance in the GB CLP Regulation, the Scientific Advisory Group on Chemical Safety (SAG-CS), the equivalent of the European SCCS, assessed the safety of the substance and has just published its conclusions.
Hexyl salicylate (CAS No.6259-76-3, EC No.228-408-6, INCI: Hexyl Salicylate) is a fragrance ingredient that has been included in the GB Mandatory Classification and Labelling (MCL) (equivalent to Annex VI of the European CLP Regulation 1272/2008).
As the industry has submitted a dossier requesting an exemption from the resulting ban on the use of the substance in cosmetic products, the SAG-CS has assessed its safety.
The conclusions of the SAG-CS
Based on the evidence available to the SAG-CS, members agreed that Hexyl Salicylate is acceptable for use in cosmetic products intended for use by adults and children aged 3 years and above at the stated concentrations:
• Hydroalcoholic-based fragrances (spray and non-spray): 2%
• All rinse-off products: 0.5%
• All leave-on products: 0.3%
• All oral care products: 0.001%
Children aged 3 years and older
Data being limited for children aged 3 years of age and above, the SAG-CS recommended that a full risk assessment should be conducted when an adequate methodology and data become available in order to fully assess the use of Hexyl Salicylate in products intended for children.
Children under 3 years of age
Based on the evidence available to the SAG-CS, members agreed …