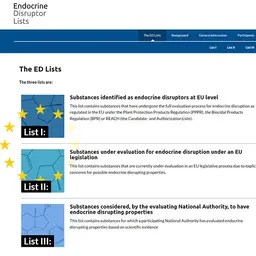
In October 2025, the SCCS accepted the European Commission’s mandate to evaluate the safety of micron-sized particulate Silver in cosmetic products. The Scientific Committee on Consumer Safety has just published its preliminary Opinion. It is open for comments until February 23, 2026.
Background
Silver (CAS/EC No.7440-22-4/231-131-3) is an ingredient primarily used as a colorant (CI 77820) in cosmetics.
The European Risk Assessment Committee (RAC) of ECHA issued in February 2023 an opinion recommending among others a “Toxic for Reproduction Category 2” classification.
In October 2023, the Commission services received a dossier to defend the safe use of micron-sized particulate Silver (CAS/EC No.7440-22-4/231-131-3) as a conditioning agent in cosmetic products.
In June 2024, the SCCS concluded in its Opinion (SCCS/1665/24) that micron-sized particulate Silver was not safe at concentrations up to 0.2% in rinse-off and 0.3 % in leave-on cosmetic products. In August 2025, the Commission services received an updated dossier focusing on dermal penetration studies aiming to demonstrate the safe use of the ingredient.
For an exhaustive background information, see the articles
• ECHA: RAC Opinions on classification of 2 “cosmetic” ingredients, CosmeticOBS, 15 June 2022
• Silver: final Opinion of the SCCS, CosmeticOBS, 8 July 2024
• Silver: new request for Opinion to the SCCS, CosmeticOBS, 10 November 2025
The SCCS Opinion
1) In light of the new data provided, the previous SCCS Opinion (SCCS/1665/24) and taking under consideration the classification as toxic for …