On 9 October 2025, the OPSS (Office for Product Safety and Standards) issued a call for data on a group of cosmetic ingredients suspected of having endocrine-disrupting properties. The aim is to have their safety assessed by the SAG-CS, the British Scientific Committee.
This call for data covers seven ingredients whose safety the OPSS wishes to assess due to their alleged endocrine-disrupting properties:
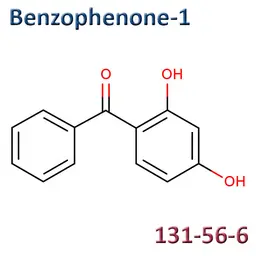
• Benzophenone-1 (CAS No.131-56-6)
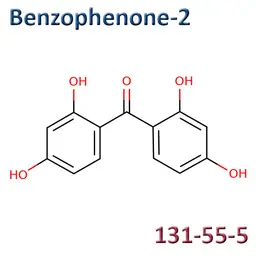
• Benzophenone-2 (CAS No.131-55-5)
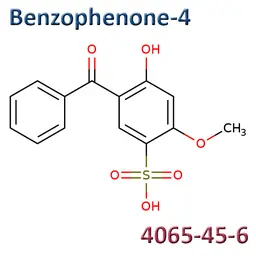
• Benzophenone-4 (CAS No.4065-45-6)
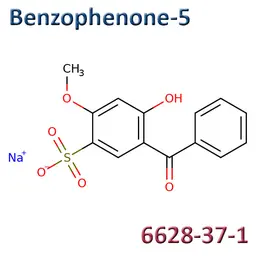
• Benzophenone-5 (CAS No.6628-37-1)
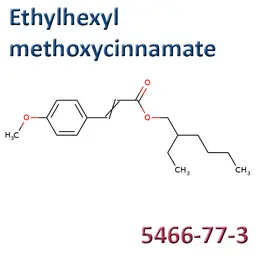
• Ethylhexyl Methoxycinnamate (CAS No.5466-77-3/83834-59-7)
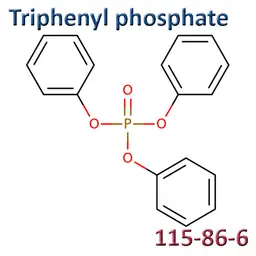
• Triphenyl Phosphate (CAS No.115-86-6)
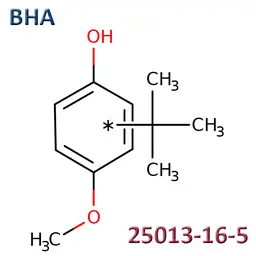
• Butylated Hydroxyanisole - BHA (CAS No.25013-16-5)
In Europe, all have already been assessed (or requested for assessment) by the CSSC and/or have recently been subject to new regulatory measures.
See the sheets for these ingredients
The OPSS points out that for its assessments, the SAG-CS (Scientific Advisory Group on Chemical Safety) will use 70 kg as the default body weight assumption for adults.
Furthermore, it specifies that if these ingredients are intended for use in products likely to be used by children or pregnant women, additional relevant data justifying their safety and exposure estimates must be provided for these specific populations.
Data may be submitted until 31 March 2025.
Source
• Call for data: safety of cosmetic ingredients, Update of 9 October 2025, OPSS, News story, 9 October 2025